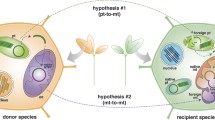
Abstract
Horizontal gene transfer (HGT) is frequent in parasitic plant mitochondria as a result of vascular connections established in host-parasite relationships. Recent studies of the holoparasitic plant Lophophytum mirabile (Balanophoraceae) revealed the unprecedented acquisition of a large amount of mitochondrial sequences from its legume host. We focused on a close relative, the generalist holoparasite Ombrophytum subterraneum, to examine the incidence of HGT events in the mitochondrial genome (mtDNA). The mtDNA of O. subterraneum assembles into 54 circular chromosomes, only 34 of which contain the 51 full-length coding regions. Numerous foreign tracts (totaling almost 100 kb, ~ 14% of the mtDNA), including 12 intact genes, were acquired by HGT from the Asteraceae hosts. Nine chromosomes concentrate most of those regions and eight are almost entirely foreign. Native homologs of each foreign gene coexist in the mtDNA and are potentially functional. A large proportion of shorter regions were related to the Fabaceae (a total of ~ 110 kb, 15.4%), some of which were shared with L. mirabile. We also found evidence of foreign sequences donated by angiosperm lineages not reported as hosts (Apocynaceae, Euphorbiaceae, Lamiaceae, and Malvales). We propose an evolutionary hypothesis that involves ancient transfers from legume hosts in the common ancestor of Ombrophytum and Lophophytum followed by more recent transfer events in L. mirabile. Besides, the O. subterraneum mtDNA was also subjected to additional HGT events from diverse angiosperm lineages, including large and recent transfers from the Asteraceae, and also from Lamiaceae.
Key message
The O. subterraneum mtDNA harbors foreign mitochondrial regions from diverse angiosperms, including 14 and 15% acquired from host lineages Asteraceae and Fabaceae. Twelve intact foreign genes coexist with native homologs.




Similar content being viewed by others
References
Alverson AJ, Rice DW, Dickinson S, Barry K, Palmer JD (2011) Origins and recombination of the bacterial-sized multichromosomal mitochondrial genome of cucumber. Plant Cell 23:2499–2513
Alverson AJ, Wei X, Rice DW, Stern DB, Barry K, Palmer JD (2010) Insights into the evolution of mitochondrial genome size from complete sequences of Citrullus lanatus and Cucurbita pepo (Cucurbitaceae). Mol Biol Evol 27:1436–1448
Arimura S, Yamamoto J, Aida GP, Nakazono M, Tsutsumi N (2004) Frequent fusion and fission of plant mitochondria with unequal nucleoid distribution. Proc Nat Acad Sci USA 101:7805–7808
Barkman TJ, McNeal JR, Lim SH, Coat G, Croom HB, Young ND, dePamphilis CW (2007) Mitochondrial DNA suggests at least 11 origins of parasitism in angiosperms and reveals genomic chimerism in parasitic plants. BMC Evol Biol 7:248. https://doi.org/10.1186/1471-2148-7-248
Bellot S et al (2016) Assembled plastid and mitochondrial genomes, as well as nuclear genes, place the parasite family Cynomoriaceae in the Saxifragales. Genome Biol Evol 8:2214–2230
Bergthorsson U, Adams KL, Thomason B, Palmer JD (2003) Widespread horizontal transfer of mitochondrial genes in flowering plants. Nature 424:197–201
Bergthorsson U, Richardson A, Young G, Goertzen L, Palmer JD (2004) Massive horizontal transfer of mitochondrial genes from diverse land plant donors to the basal angiosperm Amborella. Proc Natl Acad Sci USA 101:17747–17752
Camacho C, Coulouris G, Avagyan V, Ma N, Papadopoulos J, Bealer K, Madden TL (2009) BLAST+: architecture and applications. BMC Bioinformatics 10:421–421. https://doi.org/10.1186/1471-2105-10-421
Choi I-S et al (2019) Fluctuations in Fabaceae mitochondrial genome size and content are both ancient and recent. BMC Plant Biol 19:1–15
Cusimano N, Renner SS (2019) Sequential horizontal gene transfers from different hosts in a widespread Eurasian parasitic plant. Cynomorium coccineum Am J Bot 106:679–689
Davis C, Andersen W, Wurdack K (2005) Gene transfer from a parasitic flowering plant to a fern. Proc R Soc Lond B Biol Sci 272:2237–2242
Davis C, Wurdack K (2004) Host-to-parasite gene transfer in flowering plants: phylogenetic evidence from Malpighiales. Science 305:676–678
Davis CC, Xi Z (2015) Horizontal gene transfer in parasitic plants. Curr Opin Plant Biol 26:14–19
Delprete PG (2014) Ombrophytum guayanensis, the first record of subfamily Lophophytoideae (Balanophoraceae) in the Guayana Shield. Phytotaxa 175:263–269
Doyle JJ, Doyle JL (1987) A rapid isolation procedure for small quantities of fresh leaf tissues. Phytochem Bull 19:11–15
Edera AA, Gandini CL, Sanchez-Puerta MV (2018) Towards a comprehensive picture of C-to-U RNA editing sites in angiosperm mitochondria. Plant Mol Biol 97:215–231
Gandini CL, Sanchez-Puerta MV (2017) Foreign plastid sequences in plant mitochondria are frequently acquired via mitochondrion-to-mitochondrion horizontal transfer. Sci Rep 7:43402–43402. https://doi.org/10.1038/srep43402
Gordon D, Green P (2013) Consed: a graphical editor for next-generation sequencing. Bioinformatics 29:2936–2937
Greiner S, Lehwark P, Bock R (2019) OrganellarGenomeDRAW (OGDRAW) version 1.3.1: expanded toolkit for the graphical visualization of organellar genomes. Nucleic Acids Res 47:W59–W64. https://doi.org/10.1093/nar/gkz238
Grimes BT, Sisay AK, Carroll HD, Cahoon AB (2014) Deep sequencing of the tobacco mitochondrial transcriptome reveals expressed ORFs and numerous editing sites outside coding regions. BMC Genomics 15:31. https://doi.org/10.1186/1471-2164-15-31
Guzmán-Guzmán S (2019) A new species of Ombrophytum (Balanophoraceae), a genus not previously recorded for Colombia. Phytotaxa 424:061–066
Hansen B (1980) Balanophoraceae. Flora Neotróp 23:1–80
Hao W, Richardson AO, Zheng Y, Palmer JD (2010) Gorgeous mosaic of mitochondrial genes created by horizontal transfer and gene conversion. Proc Natl Acad Sci USA 14:21576–21581
Hepburn N, Schmidt D, Mower JP (2012) Loss of two introns from the Magnolia tripetala mitochondrial cox2 gene implicates horizontal gene transfer and gene conversion as a novel mechanism of intron loss. Mol Biol Evol 29:311–320
Kang S-H, Oh J-H, Kim H-J, Kim C-K (2017) The multipartite mitochondrial genome of Cynanchum wilfordii (Gentianales: Apocynaceae). Mitochondrial DNA Part B 2:720–721. https://doi.org/10.1080/23802359.2017.1390405
Kazama T, Toriyama. (2016). Whole mitochondrial genome sequencing and re-examination of a cytoplasmic male sterility-associated gene in Boro-Taichung- type cytoplasmic male sterile rice. PloS One
Keeling PJ, Palmer JD (2008) Horizontal gene transfer in eukaryotic evolution. Nat Rev Genet 9:1–14
Kim G, LeBlanc ML, Wafula E, dePamphilis CW, Westwood JH (2014) Genomic-scale exchange of mRNA between a parasitic plant and its hosts. Science 345:808–811
Koressaar T, Remm M (2007) Enhancements and modifications of primer design program Primer3. Bioinformatics 23:1289–1291
Kovar L et al (2018) PacBio-based mitochondrial genome assembly of Leucaena trichandra (Leguminosae) and an intrageneric assessment of mitochondrial RNA editing. Genome Biol Evol 10:2501–2517. https://doi.org/10.1093/gbe/evy179
Kubo T, Mikami T (2007) Organization and variation of angiosperm mitochondrial genome. Physiol Plant 129:6–13
Kuijt J, Delprete PG (2019) A new species of Ombrophytum (Balanophoraceae) from Chile, with notes on subterranean organs and vegetative reproduction in the family. Phytotaxa 420:264–272
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359. https://doi.org/10.1038/nmeth.1923
Larsson A (2014) AliView: a fast and lightweight alignment viewer and editor for large datasets. Bioinformatics 30:3276–3278. https://doi.org/10.1093/bioinformatics/btu531
Lowe TM, Eddy SR (1997) tRNAscan-SE: a program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res 25:955–964
Manchekar M, Scissum-Gunn K, Song D, Khazi F, McLean S, Nielsen BL (2006) DNA recombination activity in soybean mitochondria. J Mol Biol 356:288–299
Maréchal A, Brisson N (2010) Recombination and the maintenance of plant organelle genome stability. New Phytol 186:299–317
Mauseth JD, Hsiao S-C, Montenegro G (1992) Vegetative body of the parasitic angiosperm Ombrophytum subterraneum (Balanophoraceae). Bull Torrey Bot Club 119:407–417
Molina J et al (2014) Possible loss of the chloroplast genome in the parasitic flowering plant Rafflesia lagascae (Rafflesiaceae). Mol Biol Evol 31:793–803
Mower JP (2005) PREP-Mt: predictive RNA editor for plant mitochondrial genes. BMC Bioinformatics 6:96
Mower JP, Stefanovic S, Hao W, Gummow JS, Jain K, Ahmed D, Palmer JD (2010) Horizontal acquisition of multiple mitochondrial genes from a parasitic plant followed by gene conversion with host mitochondrial genes. BMC Biol 8:150
Mower JP, Stefanovic S, Young GJ, Palmer JD (2004) Plant genetics: gene transfer from parasitic to host plants. Nature 432:165–166
Nickrent DL, Blarer A, Qiu YL, Vidal-Russell R, Anderson FE (2004) Phylogenetic inference in rafflesiales: the influence of rate heterogeneity and horizontal gene transfer. BMC Evol Biol 4:40
Palmer S, Schidkraut E, Lazarin R, Nguyen J, Nickoloff J (2003) Gene conversion tracts in Saccharomyces cerevisiae can be extremely short and highly directional. Nucleic Acids Res 31:1164–1173
Park S, Grewe F, Zhu A, Ruhlman T, Sabir J, Mower JP, Jansen RK (2015) Dynamic evolution of Geranium mitochondrial genomes through multiple horizontal and intracellular gene transfers. New Phytol 208:570–583
Petersen G, Cuenca A, Moller I, Seberg O (2015) Massive gene loss in mistletoe (Viscum, Vicaceae) mitochondria. Sci Rep 5:17588
Phanstiel DH. (2019). Sushi: tools for visualizing genomics data. R package version 1.24.0.
Rice DW et al (2013) Horizontal transfer of entire genomes via mitochondrial fusion in the angiosperm Amborella. Science 342:1468–1473
Richardson AO, Rice DW, Young GJ, Alverson AJ, Palmer JD (2013) The "fossilized" mitochondrial genome of Liriodendron tulipifera: an ancestral gene content and order, ancestral editing sites, and extraordinarily low mutation rate. BMC Biol 11:29
Roney J, Khatibi P, Westwood J (2007) Cross-species translocation of mRNA from host plants into the parasitic plant doddler. Plant Physiol 143:1037–1043
Sanchez-Puerta MV (2014) Involvement of plastid, mitochondrial and nuclear genomes in plant-to-plant horizontal gene transfer. Acta Soc Bot Pol 83:317–323
Sanchez-Puerta MV, Edera A, Gandini CL, Williams AV, Howell KA, Nevill PG, Small I (2019) Genome-scale transfer of mitochondrial DNA from legume hosts to the holoparasite Lophophytum mirabile (Balanophoraceae). Mol Phylogenet Evol 132:243–250. https://doi.org/10.1016/j.ympev.2018.12.006
Sanchez-Puerta MV, García LE, Wohlfeiler J, Ceriotti LF (2017) Unparalleled replacement of native mitochondrial genes by foreign homologs in a holoparasitic plant. New Phytol 214:376–387. https://doi.org/10.1111/nph.14361
Sanchez-Puerta MV, Zubko MK, Palmer JD (2015) Homologous recombination and retention of a single form of most genes shape the highly chimeric mitochondrial genome of a cybrid plant. New Phytol 206:381–396
Sato H. (2014). Anatomía reproductiva de las especies de Lophophytum Schott & Endl. (Balanophoraceae) de la Argentina y revisión taxonómica del género en América, Universidad Nacional del Nordeste.
Sawyer S (1989) Statistical tests for detecting gene conversion. Mol Biol Evol 6:526–538
Schelkunov MI, Nuraliev MS, Logacheva MD (2019) Rhopalocnemis phalloides has one of the most reduced and mutated plastid genomes known. PeerJ 7:e7500–e7500. https://doi.org/10.7717/peerj.7500
Shearman JR et al (2016) The two chromosomes of the mitochondrial genome of a sugarcane cultivar: assembly and recombination analysis using long PacBio reads. Sci Rep. https://doi.org/10.1038/srep31533
Shedge V, Davila J, Arrieta-Montiel M, Mohammed S, Mackenzie S (2010) Extensive rearrangement of the Arabidopsis mitochondrial genome elicits cellular conditions for thermotolerance. Plant Physiol 152:1960–1970
Shimodaira H (2002) An approximately unbiased test of phylogenetic tree selection. Syst Biol 51:492–508
Skippington E, Barkman TJ, Rice DW, Palmer JD (2015) Miniaturized mitogenome of the parasitic plant Viscum scurruloideum is extremely divergent and dynamic and has lost all nad genes. Proc Nat Acad Sci USA 112:E3515–E3524
Sloan D (2013) One ring to rule them all? Genome sequencing provides new insights into the 'master circle' model of plant mitochondrial DNA structure. New Phytol 200:978–985
Sloan D, Alverson AJ, Chuckalovcak J, Wu M, McCauley D, Palmer JD, Taylor DR (2012a) Rapid evolution of enormous, multichromosomal genomes in flowering plant mitochondria with exceptionally high mutation rates. PLoS Biol 10:e1001241
Sloan D, Müller KM, McCauley D, Taylor DR, Śtorchová H (2012b) Intraspecific variation in mitochondrial genome sequence, structure, and gene content in Silene vulgaris, an angiosperm with pervasive cytoplasmic male steritility. New Phytol 196:1228–1239
Stamatakis A (2014) RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30:1312–1313. https://doi.org/10.1093/bioinformatics/btu033
Stegemann S, Bock R (2009) Exchange of genetic material between cells in plant tissue grafts. Science 324:649–651. https://doi.org/10.1126/science.1170397
Straub SCK, Parks M, Weitemier K, Fishbein M, Cronn RC, Liston A (2012) Navigating the tip of the genomic iceberg: next-generation sequencing for plant systematics. Am J Bot 99:349–364. https://doi.org/10.3732/ajb.1100335
Su H-J et al (2019) Novel genetic code and record-setting AT-richness in the highly reduced plastid genome of the holoparasitic plant Balanophora. Proc Natl Acad Sci USA 116:934–943. https://doi.org/10.1073/pnas.1816822116
Su H-J, Hu J-M, Anderson FE, Der JP, Nickrent DL (2015) Phylogenetic relationships of santalales with insights into the origins of holoparasitic Balanophoraceae. Taxon 64:491–506
Swofford D, Olsen G, Waddell P, Hillis D. (2002). PAUP*. Phylogenetic Analysis Using Parsimony (*and other methods). Version 4. Sinauer Associates, Sunderland.
Tsujimura M, Kaneko T, Sakamoto T, Kimura S, Shigyo M, Yamagishi H, Terachi T (2019) Multichromosomal structure of the onion mitochondrial genome and a transcript analysis. Mitochondrion 46:179–186
Untergasser A, Cutcutache I, Koressaar T, Ye J, Faircloth BC, Remm M, Rozen SG (2012) Primer3—new capabilities and interfaces. Nucleic Acids Res 40:e115
Varré J-S et al (2019) Complete sequence, multichromosomal architecture and transcriptome analysis of the Solanum tuberosum mitochondrial genome. Int J Mol Sci 20:4788
Westwood J, Yoder JA, Timko M, dePamphilis CW (2010) The evolution of parasitism in plants. Trends Plant Sci 15:227–235
Westwood JH, Roney JK, Khatibi PA, Stromberg VK (2009) RNA translocation between parasitic plants and their hosts. Pest Manag Sci 65:533–539
Wu Z, Cuthbert JM, Taylor DR, Sloan DB (2015) The massive mitochondrial genome of the angiosperm Silene noctiflora is evolving by gain or loss of entire chromosomes. Proc Nat Acad Sci USA 112:10185–10191
Wu Z, Sloan DB (2019) Recombination and intraspecific polymorphism for the presence and absence of entire chromosomes in mitochondrial genomes. Heredity 122:647–659
Xi Z, Wang Y, Bradley R, Sugumaran M, Marx C, Rest J, Davis CC (2013) Massive mitochondrial gene transfer in a parasitic flowering plant clade. PloS Genet 9:e1003265
Yang Z et al (2016) Horizontal gene transfer is more frequent with increased heterotrophy and contributes to parasite adaptation. Proc Natl Acad Sci U S A 113:E7010. https://doi.org/10.1073/pnas.1608765113
Zerbino D, Birney E (2008) Velvet: algorithms for de novo short read assembly using de Bruijn graphs. Genome Res 18:821–829
Zuloaga FO, Morrone ON, Belgrano MJ, Marticorena C, Marchesi E. (2008). Catálogo de las plantas vasculares del Cono Sur. Monogr Syst Bot Missouri Bot Gard, 107.
Acknowledgements
We thank A.A. Edera for help with scripts and graphs and J.D. Palmer for critical reading of the manuscript. This work was supported by Agencia Nacional de Promoción Científica y Tecnológica (Grant No PICT-0691) and Universidad Nacional de Cuyo (Grant No 06/A724) to M.V.S.P, and by NSF (Grant No 1062432) to Indiana University, which supports the computer cluster.
Author information
Authors and Affiliations
Contributions
MER and MVSP designed the study, analyzed the data and wrote the manuscript. H.S. was involved in sample collection and taxonomic identification. LEG and GP performed host and parasite DNA extraction and purification and participated in the analysis. CLG assembled the mtDNA and helped with scripts for data analyses.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Roulet, M.E., Garcia, L.E., Gandini, C.L. et al. Multichromosomal structure and foreign tracts in the Ombrophytum subterraneum (Balanophoraceae) mitochondrial genome. Plant Mol Biol 103, 623–638 (2020). https://doi.org/10.1007/s11103-020-01014-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-020-01014-x